What to Expect?
With the release of USP General Chapter <1037> Process Analytical Technology – Theory and Practice and the FDA guidelines on PAT released in July, 2025 marked a pivotal year for biotech and pharma companies to stay current with the latest advancements in PAT and integrate them into their operations.
Connecting 50+ PAT experts in pharma and biopharma, that year’s summit addressed the challenges of high initial investment costs, integration with existing systems, and data management, while exploring the rapid growth of PAT applications in biopharmaceuticals, personalized medicine, and biologics.
Attendees had the opportunity to connect with managers and directors of PAT from large pharma, top biotechs, and leading CDMOs at the forefront of process innovation as they discussed cutting-edge innovations, practical solutions, and the strategic roadmaps to successfully navigate the future of PAT in pharma and biopharma.

You Joined Your Peers to:
The Future of PAT
Benchmark your strategies against industry leaders from top pharma companies, biotechs, and CDMOs, and explore real-world solutions and cutting-edge technologies that are shaping the future of PAT.
Tech Transfer Success
Gain actionable insights to overcome tech transfer challenges for small molecule and complex therapy manufacturing processes by hearing real-world case studies on how drug manufacturers are successfully implementing PAT.
Navigating FDA PAT Guidance
Understand New FDA Guidelines on PAT for Drug Development & Manufacturing by hearing from regulatory and industry experts on how to align your strategies for smoother approvals and compliance with GMP standards.
Driving PAT ROI
Demonstrate the value of PAT to key stakeholders and gain executive buy-in by learning from top pharma and biotech companies that are demonstrating ROI through reduced batch failures, faster release timelines, and enhanced process efficiency.
Real‑Time Release
Discover how leading pharma and biotech teams are leveraging PAT to move towards Real-Time Release Testing, driving faster production timelines while maintaining high-quality standards.
Who Did You Meet?
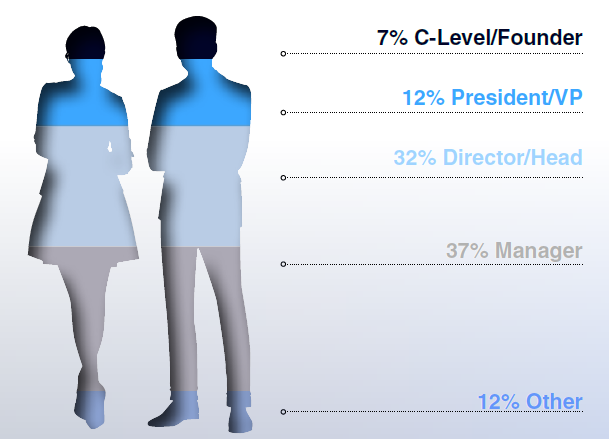
Seniority of Attendees
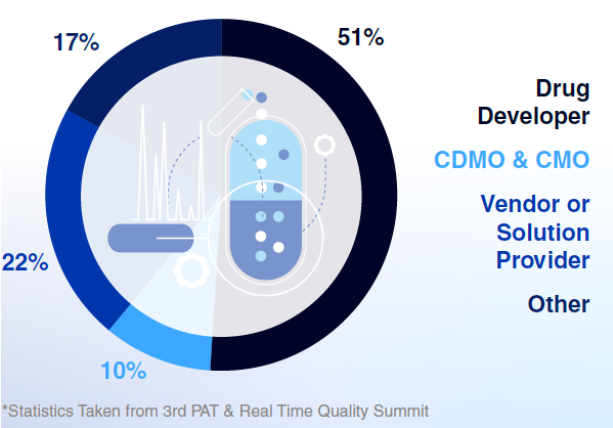
Types of Companies Attending
What Your Peers Have to Say:

Was great to have the chance to be with leading PAT practitioners and exchange and discuss about the current trends, opportunities and challenges in the pharma business. I enjoyed many high-quality talks.
Head of Advanced Analytical Technologies, Roche

The great opportunity to meet with PAT experts and share the advances and usage of PAT and chemometrics models within my company. Delighted to have learned more on other platforms such as Biotech and Cell and Gene Therapies.
Global Statistical & PAT Lead, Novartis

Well attended with representatives from companies that are highly influential in the PAT space. Small enough of a show to make networking with others comfortable. Plenty of opportunities to share thoughts with others.
Sales & Business Development Manager, Optimal Industrial Technologies

Unlike many other professional conferences I have attended, this one had a much more collaborative environment. The information was helpful, but the peer insight was unique at this event.
Senior Principal Development Engineer, Noven Pharmaceuticals




